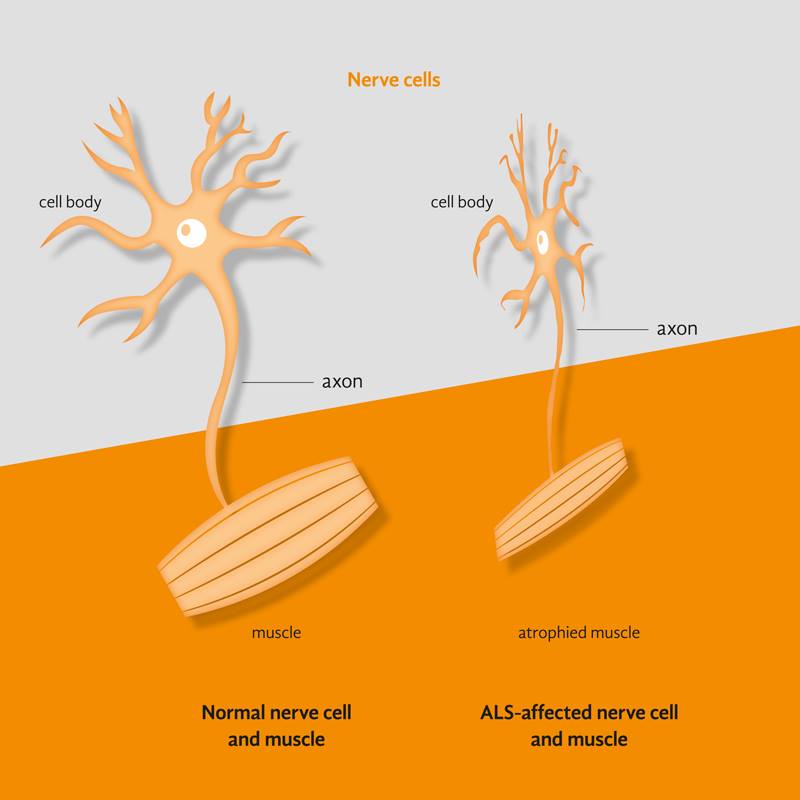
Motor Neuron DiseaseMotor Neuron Disease or MND is the name given to a group of diseases that affect special cells in the brain and spinal cord, which control the muscles. These special cells are called motor neurons and their death leads to weakness and wasting of muscles, causing loss of mobility in the limbs and/or difficulties with breathing and speech.
Amyotrophic Lateral Sclerosis (ALS) and Spinal Muscular Atrophy (SMA) are the two most common MNDs of adulthood and childhood, respectively. SMA and a small percentage of ALS cases are caused by genetic factors. In this regard, mutations or flaws in the DNA are passed from one generation to the next and the disease is said to be inherited or familial. Our laboratory is studying known and unknown genes that cause or modify MND. Several of these genes produce proteins that are expressed in every cell of the body where they perform a crucial ‘housekeeping’ role. In this regard, a key research question of our lab is why disruption of these proteins cause selective degeneration of the neuromuscular system. Knowledge gained is the first crucial step leading to discovery of effective treatments. |
Never fall in love with your hypothesis |
Are you an ALS/Motor Neuron Disease patient or relative?
Participate in our Research
Malta National ALS/MND Registry & BioBank
|
Our lab hosts the Malta's National ALS/MND Registry & BioBank, which is a unique collection of high quality ALS/MND patient samples, accompanied by detailed clinical information. This special reserve allows present and future researchers to discover novel ALS/MND causative and modifying genes through analyses of patients’ DNA, which is the blueprint of life. The identification of these genes may lead to disease pathways for which effective treatments can be developed.
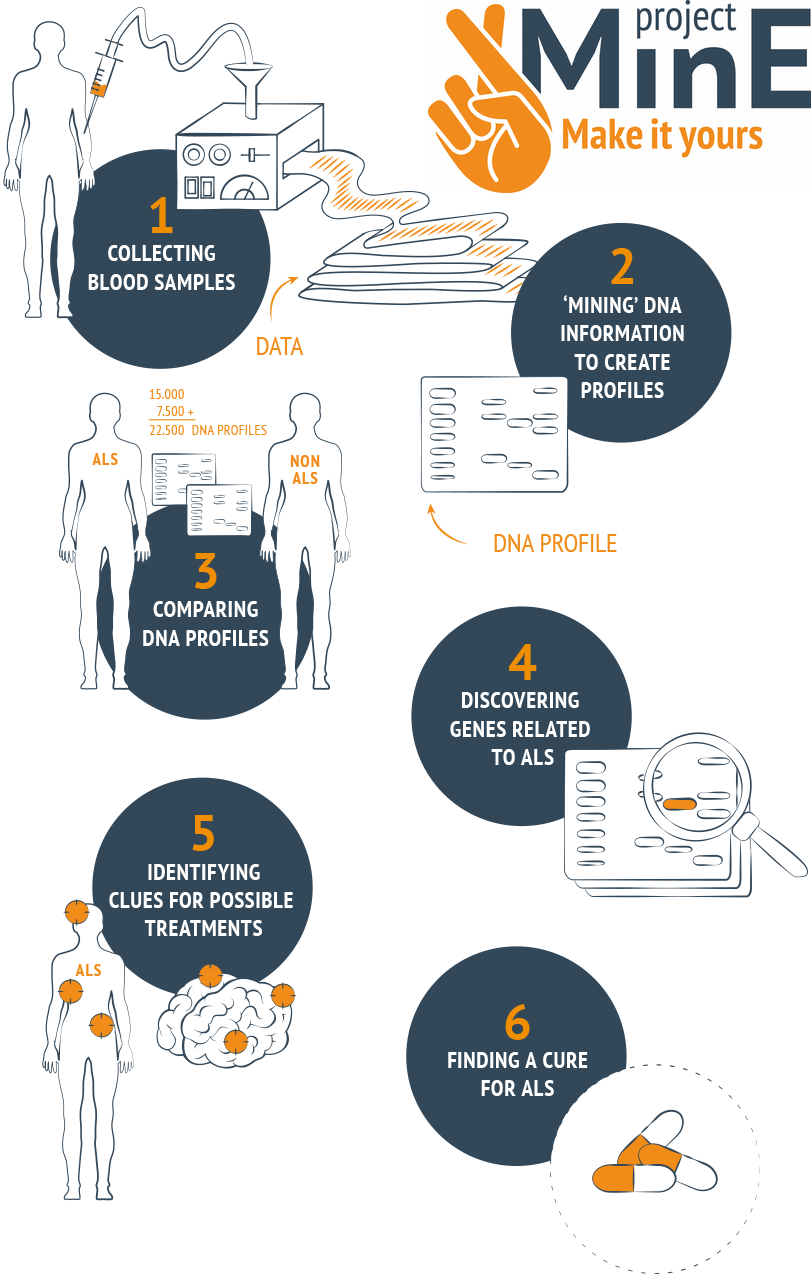
Research on samples and data gathered by Malta's National ALS/MND Registry & Biobank has revealed that Maltese ALS patients have a unique genetic makeup compared to their European counterparts (Borg et al. Eur J Hum Genet 2021). Furthermore, individuals engaged in heavy physical labour were found to have an increased risk of developing ALS, supporting a link between a history of intense physical activity and this neurological condition (Farrugia Wismayer et al. Amyotroph Lateral Scler Frontotemporal Degener 2021). Our laboratory is proudly contributing to Project MinE, which is an international groundbreaking genetic ALS research project that aims at understanding the complicated genetic basis of ALS. For successful outcomes, this research project requires enormous numbers of DNA profiles. To this end, Project MinE aims to analyse the DNA of at least 15,000 ALS patients and 7,500 controls coming from countries across the globe including Malta. If you and your relative have ALS/MND, we encourage you to be part of this global initiative. Kindly contact us for further information - confidentiality is assured. |
Mechanisms of MND
|
Our lab is at at forefront of research aimed at understanding the mechanisms of MND. Our work on the SMN-Gemins complex making use of the fly model system has allowed us to make new exciting discoveries throughout the years. Importantly, our findings point to the possibility that different MNDs share a similar disease mechanism.
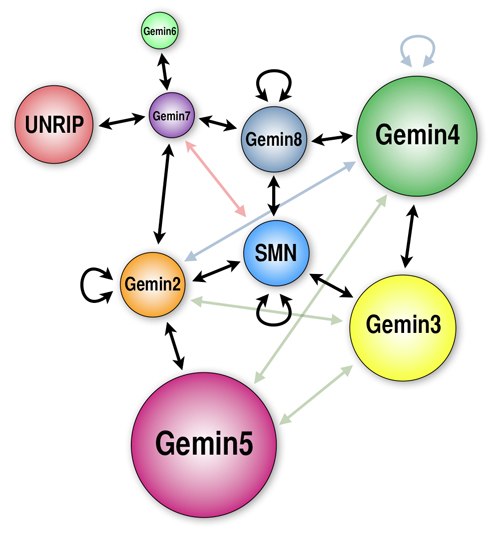
SMN-Gemins Complex The survival motor neuron (SMN) gene is the determining factor of SMA and a modifier gene in ALS. The gene produces the SMN protein, which partners with several other proteins including Gemins 2/3/4/5/6/7/8 and UNRIP to form the SMN-Gemins complex (Cauchi BioEssays 2010). Characterisation of the Gemin proteins by our lab revealed that the function of these proteins is similar to that of SMN. Thus, disruption of either SMN or the Gemins can lead to neuromuscular deficits (Cauchi et al. PLoS Genet 2008; Borg and Cauchi, PLoS ONE 2013; Lanfranco et al. FEBS Lett 2017). We also revealed that the components of the SMN-Gemins complex interact within a living model system (in vivo) and upsetting the correct levels of each component within the complex leads to catastrophic consquences that range from lethality to flies with weakened muscles (Borg et al. PLoS ONE 2015). Assembly of the splicing machinery The splicing machinery is key for cutting and pasting the cell's genetic instructions together. In an interesting study conducted by our lab, we interfered with the assembly of the splicing machinery by disrupting the cellular proteins pICln and Tgs1, two chief players in this process. Doing so, caused the fruit flies' neuromuscular system to collapse in a similar manner to patients with MND (Borg et al. Neurobiol Dis 2015). It appears that motor neurons and muscles are highly sensitive to disturbances in the assembly of the splicing machine, consequently leading to them being selectively injured (reviewed in Lanfranco et al. Front Mol Biosci 2017). Flies as Model Organisms Our research endeavours harness the rich and powerful genetic toolbox of the fruit fly Drosophila melanogaster. The fruit fly has been used in research for more than a century and has led to several landmark discoveries of which several were elevated to a Nobel Prize (1933 - Thomas H. Morgan; 1946 - Hermann J. Muller; 1995 - Edward B. Lewis, Chistiane Nusslein-Volhard & Eric F. Wieschaus; 2011 - Jules A. Hoffmann; 2017 - Jeffrey C. Hall, Michael Rosbash & Michael W. Young). In view of the extraordinary degree of genetic and physiological conservation to humans, the fly is presently regarded as an excellent animal model of neurodegenerative disease and has been successfully used to discover mechanisms or pathways of MND/ALS. Gene Discovery Our lab has embarked on a large long-term study that will reveal the complete gene-set that influences the health of the neuromuscular system. Making use of the fruit fly, we will be hitting more than 15,000 genes, one at a time, to identify those that modify the symptoms of MND. Genes discovered might have an important role in the disease mechanisms of MND. So far, we have discovered 2 key genes and by identifying their function, we earned the privilege of naming them Valette and Gaulos (Lanfranco et al. FEBS Lett 2017). Valette was chosen for the most famous Grand Master’s ‘warrior’ characteristic, considering that the gene is related to muscle strength. Gaulos – the most recognised ancient name for Gozo – actually derives from a word that means ‘vessel’. In the embryonic stage, the localisation pattern of this gene looks like a ship. Our study might take years to complete but it is expected to unveil new genes linked to MND/ALS. Some might serve as potential drug targets to halt or slow down the progressive collapse of muscle strength in MND patients. |
|
Connecting diverse ALS Genes
|
Mutations in any of an ever-increasing list of diverse genes have been identified to cause ALS with TDP-43, FUS and SOD1 featuring at the top considering that together they are responsible for a large percentage of ALS cases with a family history. Through investigations on fruit flies, our lab was able to identify a gene whose mild perturbation was enough to trigger worsening of ALS symptoms caused by disruption of TDP-43, FUS or SOD1 (Cacciottolo et al. Sci Rep 2019). The gene, named Gemin3, produces an enzyme offering researchers the possibility of tuning its function to ameliorate ALS symptoms.
Our findings point to an overlap in disease-causing mechanisms underlying each different ALS-causing gene. Right now, the research team is determining whether targeting multiple players in the pathway uncovered by Gemin3 can ameliorate ALS, a result that can potentially pave the way for development of treatments that are effective to a broad swathe of ALS patients. |
Copyright © ALS/MND LAB
Dept. of Physiology & Biochemistry
Faculty of Medicine & Surgery
Centre for Molecular Medicine & Biobanking
U N I V E R S I T Y O F M A L T A
Dept. of Physiology & Biochemistry
Faculty of Medicine & Surgery
Centre for Molecular Medicine & Biobanking
U N I V E R S I T Y O F M A L T A
The opinions and statements expressed in this website do not necessarily reflect the view of the University of Malta